Flexible, modular design with powerful visualization gives doctors a
single device with three sheath options to both diagnose and treat
patients
MARLBOROUGH, Mass.--(BUSINESS WIRE)--
Hologic, Inc. (Nasdaq: HOLX) announced today that it has received a CE
mark in Europe for its Omni™
hysteroscope, an innovative three-in-one modular scope with advanced
visualization capabilities designed for both diagnostic and therapeutic
hysteroscopic procedures. Obstetricians and gynecologists (ObGyns) can
use the new Omni hysteroscope in out- and in-patient settings.
This press release features multimedia. View the full release here:
https://www.businesswire.com/news/home/20190304005177/en/
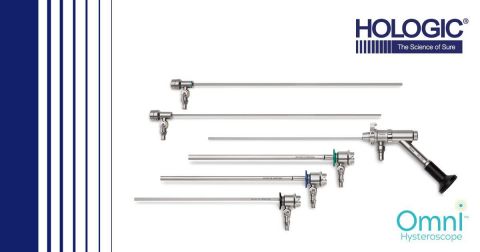
Omni™ hysteroscope (Graphic: Business Wire)
“Experts agree that direct visualization of the uterine cavity in women
with abnormal uterine bleeding is the gold standard that allows ObGyns
to accurately identify and collect quality samples and remove pathology
– in a safer and more effective manner than blind biopsy and curettage,”1
said Edward Evantash, M.D., Medical Director and Vice President of
Global Medical Affairs, Hologic. “Featuring three easily interchangeable
sheaths in one scope, our new Omni scope gives ObGyns excellent
visualization capabilities with the convenience of seeing and treating
pathology in a more streamlined procedure.”
The Omni scope is designed to help ObGyns simplify the process of
diagnosing and treating patients. Often, a surgeon requires a diagnostic
scope to look in the uterine cavity for fibroids or polyps, then
switches to an operative scope to biopsy or treat the pathology. With
the new Omni scope, ObGyns can use the same scope with different sheath
options to conveniently see and treat pathology.2
Additional features include:2
-
Premium MyoSure optics for quality visualization and clarity
throughout the procedure.
-
Sheaths designed with smaller diameters (3.7 mm diagnostic sheath; 5.5
mm operative sheath; 6 mm operative sheath) to reduce dilation
required and promote patient comfort and easy insertion.
-
Long 200 mm working length facilitates easier access and treatment for
obese patients.
-
Simple assembly and disassembly so sheaths can be changed out easily
between procedures.
-
Operative sheaths can be used with Hologic’s Aquilex™
hysteroscopic fluid management system.
-
Compatible with all MyoSure®
tissue removal offerings including MyoSure REACH, MyoSure XL, MyoSure
LITE, and MyoSure MANUAL devices.
“We continue to drive innovation designed with both our customers and
the women they treat in mind,” said Jan Verstreken, Regional President,
EMEA and Canada, Hologic. “Our customers are under pressure to deliver
high-quality care to their patients in innovative ways. Our new Omni
Hysteroscope provides them with a pioneering device that can treat a
diverse range of patients.”
About Hologic
Hologic, Inc. is an innovative medical technology company primarily
focused on improving women’s health and well-being through early
detection and treatment. For more information on Hologic, visit www.hologic.com.
Hologic, MyoSure, Omni, The Science of Sure, and associated logos are
trademarks and/or registered trademarks of Hologic, Inc. and/or its
subsidiaries in the United States and/or other countries. All other
trademarks, registered trademarks, and product names are the property of
their respective owners.
Important Safety Information
The Omni™ hysteroscope is intended to provide viewing of the cervical
canal and uterine cavity for the purpose of performing diagnostic and
operative procedures. The Omni hysteroscope is not appropriate for
patients who are exhibiting acute pelvic inflammatory disease or exhibit
the following conditions: inability to distend uterus, cervical
stenosis, cervical/vaginal infection, uterine bleeding or menses,
invasive carcinoma of the cervix, recent uterine perforation, medical
contraindications, or intolerance to anesthesia. For detailed benefit
and risk information, please consult the Instructions
for Use.
Forward-Looking Statements
This news release may contain forward-looking information that involves
risks and uncertainties, including statements about the use of Hologic
products. There can be no assurance these products will achieve the
benefits described herein or that such benefits will be replicated in
any particular manner with respect to an individual patient, as the
actual effect of the use of the products can only be determined on a
case-by-case basis. In addition, there can be no assurance that these
products will be commercially successful or achieve any expected level
of sales. Hologic expressly disclaims any obligation or undertaking to
release publicly any updates or revisions to any such statements
presented herein to reflect any change in expectations or any change in
events, conditions or circumstances on which any such data or statements
are based.
This information is not intended as a product solicitation or promotion
where such activities are prohibited. For specific information on what
products are available for sale in a particular country, please contact
a local Hologic sales representative or write to womenshealth@hologic.com.
References:
|
1.
|
|
Salim S, et al. Diagnosis and Management of Endometrial Polyps: A
Critical Review of the Literature. J Minim Invasive Gynecol,
2011;18(5):569-581
|
|
2.
|
|
Omni hysteroscope Instruction for Use. October 2018.
|
|
|
|
SOURCE: Hologic, Inc.

View source version on businesswire.com:
https://www.businesswire.com/news/home/20190304005177/en/
Media Contact:
Jane Mazur
+1 508.263.8764 (direct)
+1
585.355.5978 (mobile)
jane.mazur@hologic.com
Investor Contact:
Michael Watts
+1 858.410.8588
michael.watts@hologic.com
Source: Hologic, Inc.